Authors: Frédéric Thomas, Antoine M. Dujon
Source: BioEssays (Dec 2025)
Abstract
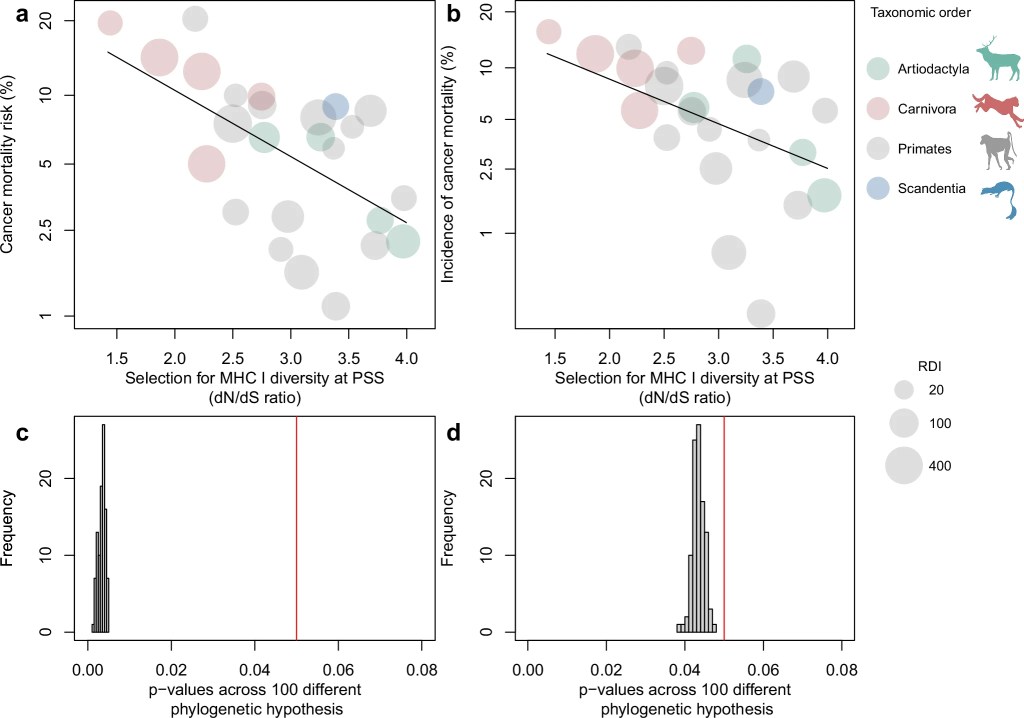
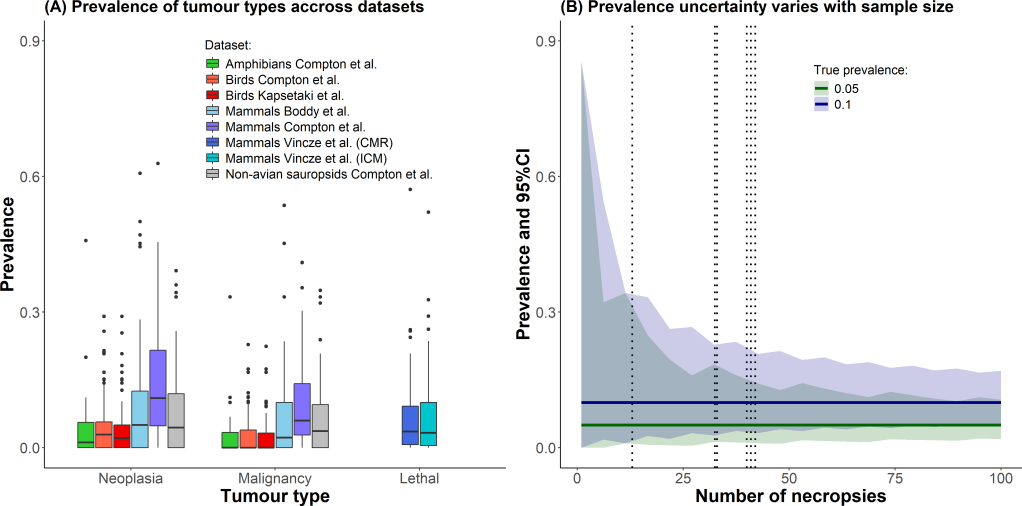
Laplane et al. recently provided a valuable framework for understanding cancer evolution through multilevel selection (MLS), distinguishing between MLS1, where groups differ in persistence based on the traits of their constituent cells but do not reproduce or evolve group-level adaptations, and MLS2, where groups themselves reproduce and possess emergent fitness distinct from that of individual cells. However, as the authors themselves acknowledge, applying MLS2 to metastasis is challenging for several reasons.
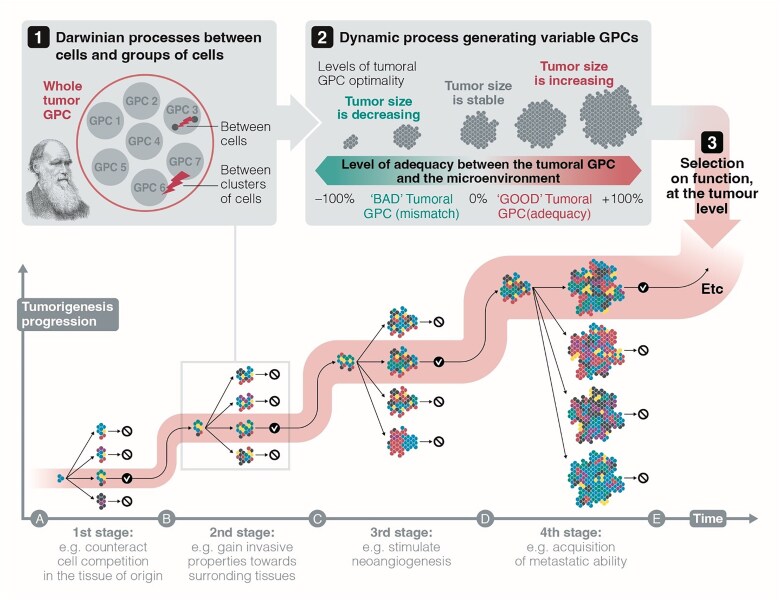
We argue that, rather than behaving as isolated evolutionary units, tumor sites function as components of a distributed system. This perspective suggests that metastasis may be better understood through the lens of selection for function, a framework that explains how traits contributing to system-level persistence can be maintained without requiring group-level reproduction.
This approach complements MLS theory and helps account for the resilience of the metastatic system as a whole, namely, the persistence and coordination of multiple tumor sites functioning as a collective rather than as isolated tumors, beyond classical Darwinian models.
It also aligns with the view that metastasis may reflect the reactivation of ancient cellular programs in a novel, nonreproductive context.